The path of the pandemic and the associated public health restrictions will be the most important near-term determinants of the UK’s economic and fiscal performance. Our March 2021 central forecast was conditioned on the Government’s ‘Roadmap’ for the lifting of restrictions and despite encouraging news regarding vaccines, there remains considerable uncertainty surrounding the future path of the pandemic and the economy. This box considered the uncertainties around key epidemiological assumptions on how the pandemic will unfold.
This box is based on Imperial College London, Unlocking Roadmap Scenarios for England and GOV.UK data from February 2021 .
The Roadmap and our central forecast assume that the vaccine rollout will deliver a substantial reduction in virus-related morbidity and mortality. The epidemiological modelling carried out by the Government’s public health advisors and academics at the University of Warwick and Imperial College, London show just how much of an improvement is possible while lifting the bulk of public health restrictions.a But it also demonstrates the sensitivity of hospitalisations and deaths to small changes in the transmissibility of the virus, vaccine effectiveness, and individuals’ behaviour. This reflects the exponential nature of the spread of the virus, especially the new Kent variant which accounts for the majority of cases in the UK.
Given the unpredictability of the virus and the novelty of the vaccines, there is still considerable uncertainty as to how the pandemic will unfold henceforth. Key sources of uncertainty include:
- Vaccine rollout. Modelling by the Government’s public health advisors assumes a rollout speed reaching 4 million per week from 25 April, continuing at 3.9 million per week in May before being sustained at this level in a fast rollout scenario.b The Government plans to have offered first doses of vaccines to all priority groups (covering 32 million people) by 15 April and all adults (53 million) by 31 July.c If the early February rollout speed of around 3 million doses a week were maintained, these targets should be met. But the quicker the vaccine rollout, the sooner transmission, hospitalisations and deaths can be reduced. If the rollout speed increased further to 4 million a week from March onwards, all adults could receive their first dose by June.
- Vaccine take-up. The modelling assumes take-up of 95 per cent for over 80s, 85 per cent for 50 to 80-year olds and 75 or 85 per cent for under 50s in their central scenarios.d
- Surveys conducted at the end of 2020 suggested that some people could be reluctant to be vaccinated.e However, take-up among the top priority groups has so far been very high. As of mid-February, over 90 per cent of over-75s in the UK had been vaccinated.f This suggests that take-up across the population could be higher than expected. But takeup may still be low for groups who perceive the risks from infection to be low, including the young, or who distrust the public authorities. This could result in clusters of infections, hospitalisations and deaths that require localised restrictions to control.
- The effectiveness of vaccines in reducing hospitalisation and deaths. Consistent with evidence from clinical trials, the modelling assumes vaccines reduce the risk of hospital admission and death by 70 to 86 per cent after one dose and by 80 to 98 per cent after two doses. Typically, however, the impact of vaccines in the community is less than in trials, so actual effectiveness could be lower. That said, early evidence suggests that effectiveness in the community is at least as good as in trials. For example, early data analysed by Public Health England suggests that hospitalisation and death from the virus will be reduced by over 75 per cent in those who have received one dose of the Pfizer vaccine.g
- The impact of vaccines on virus transmission. The modelling assumes vaccines reduce the risk of becoming infected (as opposed to suffering serious illness) by 48 to 65 per cent after one dose and by 60 to 94 per cent after two doses. This is a crucial assumption because it determines the extent to which the vaccinated can still spread the virus to the unprotected. Initial studies suggest that a single dose of the Oxford vaccine reduces the acquisition of infection (with or without symptoms) by 64 per cent.h A University of Warwick study found that under relatively pessimistic assumptions about vaccine take-up, even an 85 per cent transmission reduction may be insufficient to keep the reproduction rate of the virus, R, below 1 once restrictions are fully lifted.i
- Duration of immunity. Modelling (which is typically over a relatively short time horizon) generally assumes full immunity persists. But declining immunity is observed after infection by other coronaviruses and a small number of recovered individuals are known to have been reinfected indicating that recovery does not guarantee subsequent immunity.j A UK Biobank study of over 20,000 individuals who had tested positive found that while 99 per cent of participants showed antibodies three months after infection, that had declined to 88 per cent after six months.k This study did not consider the duration of immunity as a result of vaccinations. Given the first vaccinations were given in real world conditions in the UK in December 2020, data on the duration of immunity from vaccination will take more time to accrue. It is not unreasonable, however, to expect people to suffer periodic reinfections before they are vaccinated and to need regular booster vaccinations.
- New virus variants. The modelling assumes no impact of novel variants other than the Kent variant, so the possibility of further new variants represents a key downside risk. Were a new variant to be resistant to existing vaccines, they would need to be reconfigured – which can be expected to take several months – and a new vaccine programme rolled out (much as happens annually with influenza vaccinations). In the interim, such a new variant would result in additional hospitalisations and deaths unless offset by the reimposition of public health restrictions. Early evidence suggests that existing vaccines remain effective against the Kent variantl but that the existing Oxford vaccine may be less effective against the South African variant.m The emergence of new variants is more likely the higher the prevalence of the disease. Rapid and effective testing and isolation are both necessary to slow their spread.
- Seasonality of infections. The modelling generally abstracts from seasonality, though it is considered in sensitivity analysis. The full extent of seasonality for coronavirus is not yet clear, although more social interaction takes place indoors in the winter and there is evidence that the virus is less transmissible in the summer.n That suggests a seasonal pattern is likely, as with other respiratory illnesses, such as influenza.
- Compliance with public health restrictions. The modelling usually avoids explicit assumptions about compliance with public health restrictions. Lower compliance would reduce the effectiveness of a given set of restrictions in reducing transmission. It seems likely that compliance will decline once a significant fraction of the population has been vaccinated. Evidence from vaccine rollouts for Lyme disease and influenza in the USA found that adherence to rules indeed fell.o And a December 2020 YouGov survey found nearly a third of respondents said they would be less strict in sticking to the rules after being vaccinated while one in ten would ‘probably no longer follow the rules’ at all.p
- New treatments and/or therapeutics. The modelling assumes no impact from new treatments or therapeutics, drawing on past data in setting infection-hospitalisation and infection-fatality rates. So successful new treatments could have a material beneficial effect on future levels of hospitalisation and death for a given level of infection. More importantly for our forecasts, if the ‘fear factor’ associated with the virus was to be largely removed for most of the population, economic activity could return closer to pre-virus norms. Randomised trials in 2020 found that steroid treatments can reduce deaths by up to one third in hospitalised patients and trials of other treatments are under way.q
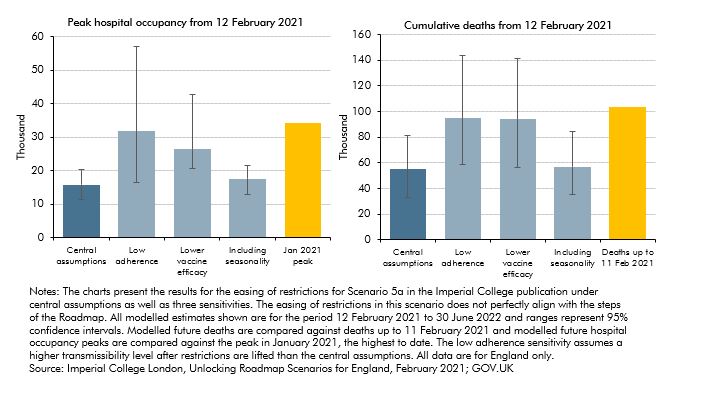
The Government’s modelling considered the epidemiological risks posed by several of these assumptions, including: low adherence to restrictions; lower vaccine efficacy; and seasonality. Chart A displays results from epidemiological modelling by Imperial College, London for an easing scenario similar to the Roadmap, under central assumptions and alternative assumptions. Results for potential future deaths and hospital occupancy peaks are compared against past deaths and hospital occupancy peaks. The Chart shows that more pessimistic assumptions about either adherence to restrictions or vaccine efficacy could result in around 95,000 deaths over the period from February 2021 to June 2022, around 40,000 more than under central assumptions. Future hospital occupancy peaks are kept well below the January 2021 peak under central assumptions but more pessimistic assumptions about adherence or vaccine efficacy could result in peaks of similar magnitudes to January 2021.
Chart A: Potential future epidemiological outcomes as restrictions are eased compared to past outcomes
This box was originally published in Economic and fiscal outlook – March 2021
